A single shot transforms the mice’s brains right into a biomanufacturing machine. Blood proteins churn the injected chemicals right into a soft, flexible electrode mesh that seamlessly wraps around delicate neurons. Pulses of sunshine aimed toward the mesh quiet hyperactive cells. All of the while, the mice go about their merry ways, with no inkling they’ve been become cyborgs.
This science fiction-like invention is the brainchild of Purdue University scientists in search of to reimagine brain implants.
These devices, often composed of rigid microelectrode chips, have already modified lives. They will collect electrical signals from the brain or spinal cord and translate these signals into speech or movement—returning lost abilities to individuals with paralysis or diseases of the brain. Implants may also jolt brain activity and pull people out of severe depression.
Yet most implants require extensive surgery and risk damaging the brain’s delicate tissue. The brand new technology would avoid these downsides by constructing electrodes directly on the goal.
“Our work points to a future where doctors could ‘grow’ soft, wire-free electronic interfaces contained in the brain using the patient’s own blood, then gently dial brain activity up or down from outside the pinnacle using harmless near-infrared light,” study writer Krishna Jayant said in a press release.
Probes Galore
The brain produces every certainly one of our sensations, movements, emotions, and decisions. Scientists have long sought to decode and manipulate its activity with a spread of hardware.
Some devices use electrodes to watch single neurons in a lab dish. Others are physically inserted into brain regions that encode cognition and emotion. Some designs sit atop the brain, without puncturing its delicate tissue, and capture dynamic brain waves like a wide-lens camera.
But brain tissue is soft and squishy; microelectrodes will not be. The mismatch often results in scarring, signal loss, and shortened device lifetimes. Replacing broken or infected implants is surgically complex and may further damage the brain. Some experts have even raised ethical concerns about long-term care.
A recent explosion of sentimental, biocompatible materials suggests alternatives are possible, and we’ve seen a wave of creative recent probes. In a single example, a silk-like mesh drapes over the brain’s surface, and a related version maps electrical activity in brain organoids. One other device is smaller than a cell and, after injection, hitches a ride on immune cells into the brain. These systems can record and alter brain activity. But prebuilt implants often require surgery and struggle to integrate with their hosts without damaging surrounding tissue.
So, why not grow an electrode directly contained in the brain?
“The flexibility to synthesize [conductive] materials on demand at a goal site could overcome the restrictions of conventional synthetic implants,” wrote M.R. Antognazza and G. Lanzani on the Italian Institute of Technology, who weren’t involved within the study.
Under Construction
Our cells are natural manufacturers, always assembling things like proteins, genetic messengers, and membranes. Cells depend on two essential ingredients to construct the complex structures of life: Biological constructing blocks and catalysts to bind them together. Synthetic materials work the identical way. Monomers link like Lego blocks to form polymers with the assistance of a catalyst.
The invention of electrically conductive polymers, meanwhile, has galvanized efforts to grow living bioelectronics directly contained in the body. In a previous study, researchers genetically engineered cells to supply a protein catalyst that helps assemble conductive structures on the surfaces of living neurons. One other approach used hydrogen peroxide—a standard first-aid staple—to compile monomers into reliable electrodes that monitor nerves in leeches.
These quirky early successes showcased the promise of brain-built electronics, but hit hard limits. The chemistry often relied on catalysts toxic to neurons. Even when successfully formed, the electrodes mostly just listened. Changing brain activity required additional physical cables.
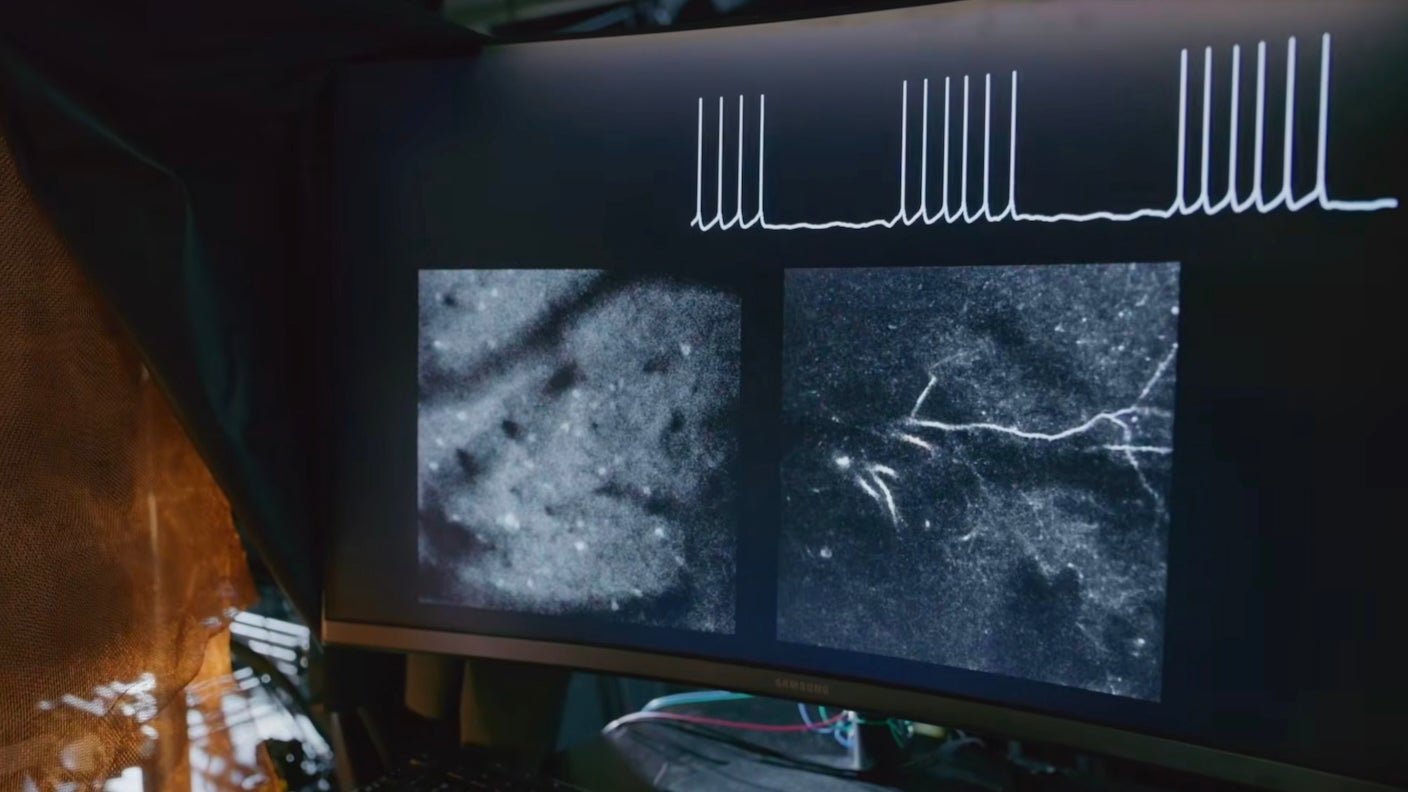
The Purdue team rewrote the recipe. They designed a monomer, called BDF, that with the assistance of hemoglobin—a protein in red blood cells—becomes a soft, flexible, and electrically conductive mesh surrounding neurons at the positioning of injection. The willowy electrode hugs the brain’s anatomy and moves with it, minimizing physical damage. It’s attentive to near-infrared light and may translate light pulses from outside the skull into electrical signals that alter brain activity.
“Our key idea was to let the body’s own chemistry do the exertions,” said study writer Sanket Samal.
The appeoach worked in several tests. Injecting BDF into store-bought beef and lamb steaks produced the electrode mesh inside a day at human body temperature. In zebrafish embryos, a darling in neuroscience research, the response proceeded easily inside their yolks. Over 80 percent of the embryos survived, developed normally, and actively swam around—suggesting minimal harm.
But steak dinners and translucent fish are a far cry from our brains. Mice are closer. With the assistance of blood, BDF formed electrodes in mice’s motor cortexes after injection with minimal surgery. The mice’s brains maintained a standard balance of activity as they skittered around.
The team also coaxed dendrites, the tree-like input branches of a neuron, to supply the conductive mesh. Dendrites aren’t just passive cables, they’re “mini computers” that contribute to the brain’s computation and learning. Current methods struggle to exactly single out and control dendrite activity without messing with other parts of the neuron.
With near-infrared light, dendrite-built electrodes modified the best way the neural branches behaved. The sunshine temporarily lowered brain activity, and mice trained to press a lever were unable to perform the duty. It didn’t wipe out their memory though: After turning off the sunshine, the animals regained the skill. Their brains showed no signs of infection, inflammation, or over-heating throughout the study.
Inhibiting brain signals has upsides. Hyperactive brain activity in epilepsy and Parkinson’s disease, for instance, is currently dampened with medication or—in severe cases—brain implants. If validated, brain-grown electrodes might be a less invasive alternative. Though to be clear, the tactic still requires surgery to inject the materials. Adding biocompatible magnetic ingredients, which may also control brain activity, could further boost the system’s potential.
How long the materials stay put and in the event that they’re protected over the long run stays unclear. But in theory, the strategy could also control spinal cord nerves or heart tissue. Researchers could also adapt the technique to use other sorts of materials that regulate brain activity in alternative ways, like ramping it up.
With further improvement, the electrode wouldn’t “just coexist with brain cells for months or years; it becomes a part of them, stable across lifetimes,” said Jayant.